< Back
Intensifier Control
Intensity-based FRET
Forster Resonance Energy Transfer (FRET) efficiency E indicates the percentage of the excitation photons that contribute to FRET and is defined as:
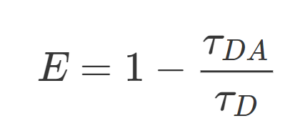
where τDA is the fluorescence lifetime of the donor in the presence of an acceptor, and τD in the abscence of an acceptor. As you can see, the more FRET occurs, the more decrease in donor fluorescence lifetime.
FRET strongly depends on the distance between the donor and acceptor fluorophores (sixth-power relationship). Fluorescence lifetime of a fluorescent molecule is inversely proportional to its FRET efficiency, thus the higher the FRET efficiency the lower the fluorescence lifetime of the donor molecule will be.
The efficiency also depends on the donor-to-acceptor separation distance R with an inverse 6th order law due to the dipole-dipole coupling mechanism:
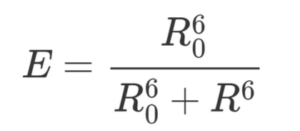
with R being the distance between donor and acceptor pair and R0 being the Förster distance between donor and acceptor at which the FRET efficiency is 50%.
FRET efficiency in a single pixel of an image, does not give exact conclusions about the interactions between fluorophores. The entire 2D image gives a better overview of the interactions that occur. For example: in case of 50% FRET efficiency in a single pixel, it could be possible that 50% of the donor fluorophores have had 100% energy transfer to acceptor fluorophores, but it also could be possible that 100% of the donor fluorophores have had 50% energy transfer to acceptor fluorophores.
Fluorescence imaging Cameras

HiCAM Fluo
The HiCAM Fluo is a high-speed camera for fluorescence imaging applications. It records high resolution images at a frame rate of up to 2000 fps in the most challenging low-light conditions by using a cooled image intensifier. Packed into a compact aluminum enclosure, it is easy to attach the HiCAM Fluo to any fluorescence microscope.
View Product
TRiCAM
The TRiCAM is a compact intensified camera. It is designed for scientific and industrial applications that require low-light imaging. With built-in signal generators, the TRiCAM is capable of ultra-short exposures through fast gating and therefore suitable for time-resolved imaging.
View Product
TRiCATT
The TRiCATT is a compact lens-coupled image intensifier for scientific and industrial applications that require: – Low-light level imaging – Ultra-short exposures through fast gating – Frequency-domain imaging using lock-in detection Any camera with C-mount and a 1/2″, 2/3″, or 1″ image sensor is compatible with the TRiCATT. You can find the right TRiCATT for your camera with our interactive calculator.
View Product
