< Back
Intensifier Control
Fluorescence Lifetime Imaging Microscopy
(FLIM)
What is Fluorescence Lifetime Imaging Microscopy (FLIM)
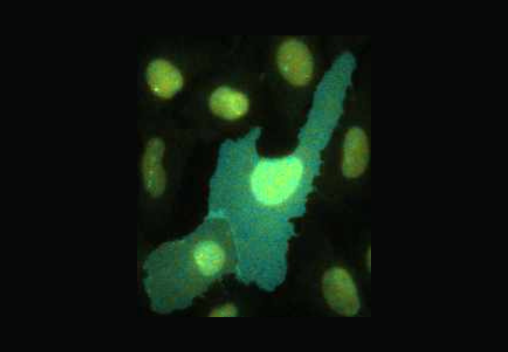
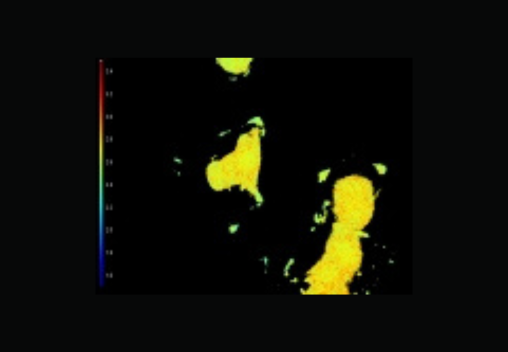
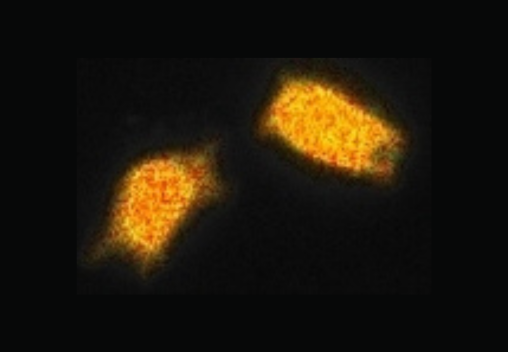
FLIM has become an important tool to assess the biochemical environment of fluorescent molecules and probes.
Upon excitation, fluorescent molecules emit light and the fluorescent lifetime quantifies the decay rate of that emitted light. The fluorescence lifetime is a telltale signature of the molecules and their immediate environment.
FLIM is the technique which maps the spatial distribution of lifetimes in living cells and in inorganic material. Fluorescence lifetime is independent of concentration, bleaching and intensity variations, making it an inherently quantitative technique, and a key advantage over the light intensity.
FLIM for Beginners Application Note
Unlock a new dimension in fluorescence imaging with FLIM for Beginners. Unlike intensity-based methods, FLIM measures fluorescence decay—revealing insights into biochemical and biophysical environments. This guide covers the fundamentals, the two main approaches (Time and Frequency Domain), and Phasor Analysis, plus key applications like metabolic imaging, protein interactions, and fluorophore unmixing.
Download it to strengthen your FLIM knowledge and enhance research in cancer, neuroscience, and drug discovery.

Application Notes
Cameras for FLIM

LIFA FLIM
The Lambert Instruments LIFA system is the fastest and easiest way to perform Fluorescence Lifetime Imaging Microscopy (FLIM).
View Product
TRiCAM
The TRiCAM is a compact intensified camera. It is designed for scientific and industrial applications that require low-light imaging. With built-in signal generators, the TRiCAM is capable of ultra-short exposures through fast gating and therefore suitable for time-resolved imaging.
View ProductUser Publications
Researchers around the world use the LIFA in their studies. Opposite is an overview of the most recent publications describing research that was done using a LIFA.
For a complete overview of applications, please visit our applications pages and our selected LIFA publications on the Lambert main site for more information.
Mutant APC reshapes Wnt signaling plasma membrane nanodomains by altering cholesterol levels via oncogenic β-catenin
July 19 – FLIM
Electrocatalytic Reaction Driven Flow: Role of pH in Flow Reversal
Mar 2, 2022 – FLIM, proton concentration, University of Twente
Bioorthogonal labeling of transmembrane proteins with non-canonical amino acids unveils masked epitopes in live neurons
Dec 7, 2021 – Microscopy, Fluorescence
Quantification of PD-1/PD-L1 Interaction between Membranes from PBMCs and Melanoma Samples Using Cell Membrane Microarray and Time-Resolved Förster Resonance Energy Transfer
Dec 7, 2021 – FRET, Fluorescence
Glycerol suppresses glucose consumption in trypanosomes through metabolic contest
Aug 13, 2021 – FLIM
PIM kinases inhibit AMPK activation and promote tumorigenicity by phosphorylating LKB1
Jun 30, 2021 – FLIM